Attempting to eradicate all pests from my plant collection using CO2
Table of Contents
Objective
Get rid of pests in my collection of cacti (and a few other plants), mostly seedlings, using CO₂.
Thrips
I have at least two species of thrips. One of them I believe to be F. occidentalis, the other I’m not sure. They are darker, and could be E. americanus, “greenhouse thrips”, see https://www.koppert.ca/news-information/news/canadian-thrips-in-greenhouses-grow-rooms/
The F. occidentalis have been mostly under control thanks to the Orius population I’ve been able to sustain in a grow tent with constantly blooming peppers (as in, Capsicum) as “banker plants”, see my other article Predatory Bugs.
They did make some cosmetic damage to a bunch of cactus seedlings, and I’d like to get rid of them for good (predatory arthropods usually work less well in the lower humidity required by succulent plants).
Root mealybugs
My main nuisance. Most of my 2024 and 2025 batches of cactus sowings have been infested. I’ve tried hot water treatment and soaking the roots in isopropyl alcohol, but it is very tedious and time-consuming.
I want to see if CO₂ could be a solution for large scale treatment, with acceptable losses. My priority is to get rid of the pests so that future plants are not immediately infested, not to make sure all my plants survive.
Disclaimers
- This is not proven to work, just me messing around
- I have no qualifications that should make you trust me
- Do do try this at home in hope that will get rid of your plants’ pests.
- This is probably dangerous for plants as currently described
- See next “Safety” section
Safety
CO₂ is an invisible and odorless gas, and poisoning does not give much warning: be careful if you try to replicate my experiments. Only do it outside, do not vent the box indoors.
If you have bring the box inside after it has been filled with carbon dioxide, make sure the room is well ventilated and use a sensor (near the ground) so you are warned in case of leaks. Keep pets and children away.
Keep your face away from the box when opening it.
Ideally, do not try any of that at home. It’s not currently proven to work anyway.
Target parameters
| Parameter | Target |
|---|---|
| CO₂ concentration | ≥60% |
| Temperature | 30°C |
| Duration | 24 hours |
Pest-specific resources
This table is a rough evidence ranking, not a guarantee that the treatment will work in a home setup.
| Pest | Evidence for ~60% CO₂ / 30°C / 24h | Notes | Refs |
|---|---|---|---|
| Spider mites (two-spotted, all stages incl. eggs) | Good published support | 60% CO₂ at 30°C was reported as 100% lethal to all tested stages on strawberry runners. That does not prove safety on all houseplants. | [Held et al. (n.d.); Oyamada and Murai (2013)] |
| Thrips (adults + larvae) | Good published support | Five adult thrips species showed complete mortality under 60% CO₂, with stronger results at higher temperature. 30°C is better supported than 25°C. | [Seki and Murai (2012a); Held et al. (n.d.)] |
| Thrips eggs (in plant tissue) | Good published support | Eggs inside plant tissue were killed in temperature-dependent trials at 60% CO₂. The strongest support is near 30°C. | [Seki and Murai (2012b)] |
| Broad mites / flat mites | Unclear / experimental | I did not find a direct live-plant CO₂ source for these pests during this review. | - |
| Root mealybugs | Unclear / experimental | Main uncertainty: whether lethal gas reaches pests throughout the root ball and substrate. I did not find a direct CO₂ source for them during this review. | - |
| Aerial mealybugs | Unclear / probably difficult | Mealybugs hide in sheltered spots and have waxy coverings, but I did not find a direct CO₂ source for them during this review. | - |
| Fungus gnats | Weak support | Soil-dwelling larvae were more tolerant than the other greenhouse pests in one modified-atmosphere study. | [Held et al. (n.d.)] |
Phase 1: cheap, small-scale experiment
Supplies
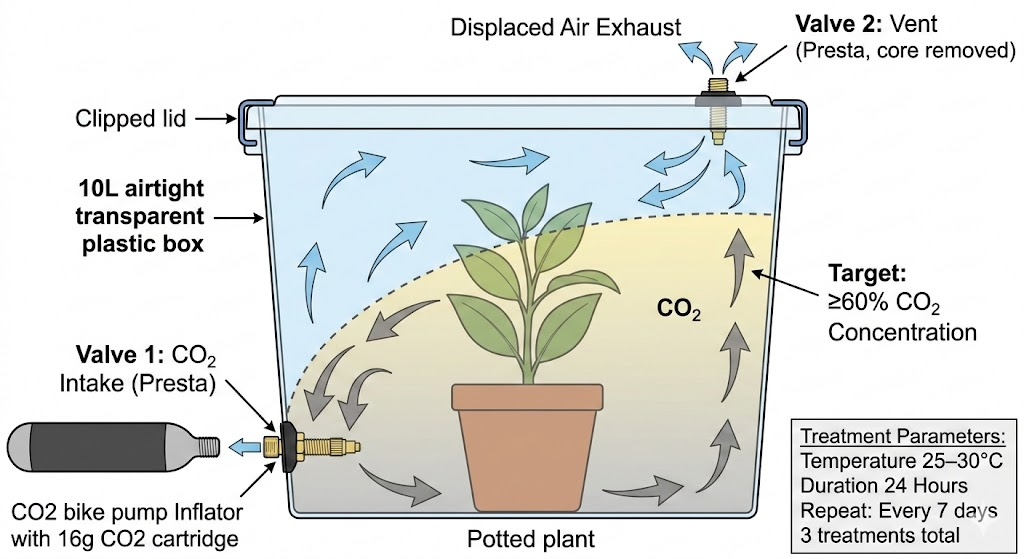
- Airtight 10L box
- 2× Presta Tubeless Tire Valve
- CO₂ bike pump
- 16g CO₂ cartridges
- conical drill bit
General idea
One 16g CO₂ cartridge produces around 8.9L of CO₂ gas at 25°C. In a 10L container, assuming some loss from the imperfect displacement, the concentration should be above 60%. Note that consumer CO₂ sensors won’t measure CO₂ concentration above 10000 ppm, so we can’t use those to check that we get the correct values.
The idea is to release CO₂ into the bottom of the box. Being heavier than air, it will fill the bottom first, and push the air out through a vent on top.
One tubeless tire valve inserted at the bottom of the box will serve as the intake, and another will serve as the vent. They come with O-rings and locknuts to make the holes airtight.
Use the bike pump to release CO₂ into the box with the air vent open, then close the vent. Now wait for 24 hours and pray that this whole idea was not doomed to fail because I’ve missed some obvious reasons for it to fail.
Issues
Unverified CO₂ concentration
My Aranet4 sensor can’t measure above 10000 ppm (1% concentration).
CO₂ sensors for measurements from 0 to 100% are a bit expensive, but maybe I could get a O₂ sensor for cheaper and use the O₂ concentration as a proxy…
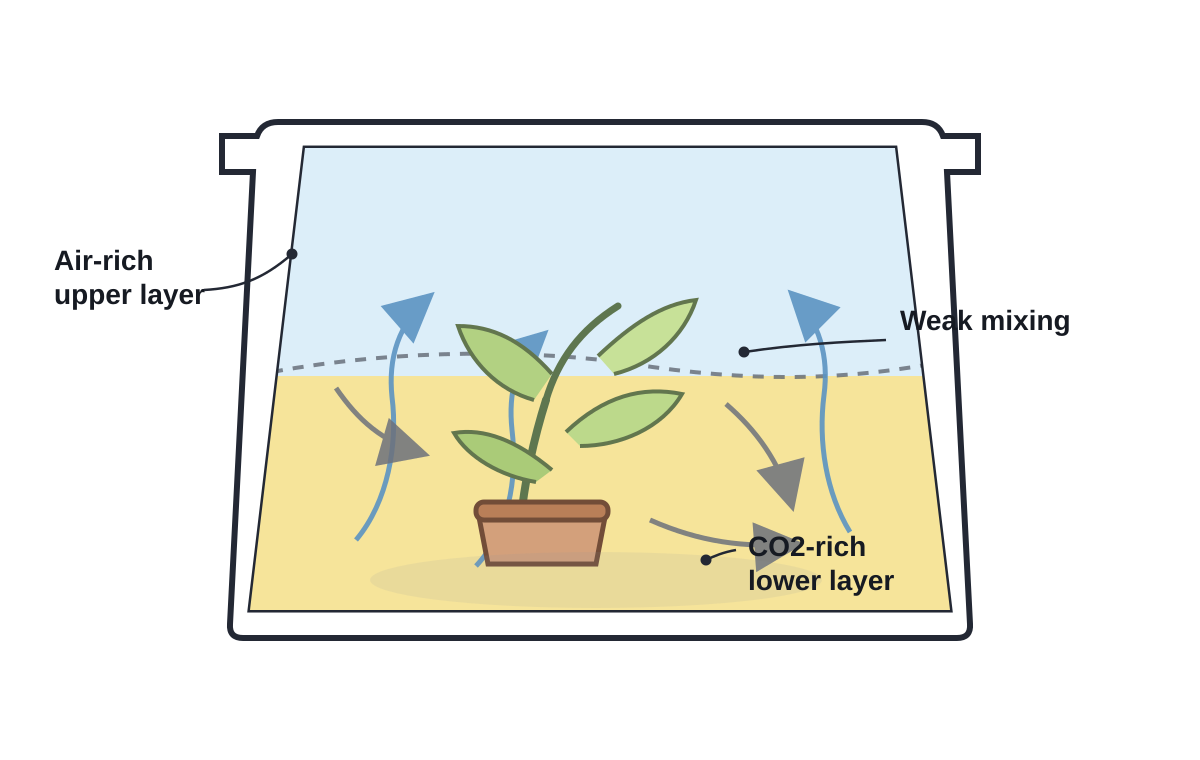
Poor gas mixing and stratification
CO₂ being denser than air, it will probably stratify.
A small fan should be added to the box, and be turned on after it is closed.
Leaking
Will the box leak? Only accurate measurement in the box could tell me that.
Hypoxia harmful to plants?
Low O₂ concentration might be as much of a danger to plants as high CO₂.
Questions
- How will I check that the box is airtight enough? Should I try first with a small amount of CO₂ and use my CO₂ sensor to check that the value is stable?
- Will I get 100% CO₂ at the bottom and <1% at the top of the box? How
to make the whole atmosphere of the box actually 60% CO₂?
- Maybe a small fan?
Assembly
- Drill a 7mm hole in the side wall, a few cm from the bottom → insert Valve 1 (CO₂ input) from inside, tighten locknut on outside
- Drill a 7mm hole in the lid, ideally on the opposite side from Valve 1 → insert Valve 2 (vent) from inside, tighten locknut on outside
- The rubber base of each valve compresses against the plastic to form an airtight seal
Note: drilling into plastic might break it, that’s why I will be using conical drill bits.
Treatment procedure
- Place plant inside the box, close and clip the lid
- Remove the valve core from Valve 2 (vent) using the included tool — this opens it as a vent hole
- Screw a 16g cartridge into the bike inflator, attach the inflator to Valve 1
- Slowly release CO₂ over 30–60 seconds using the regulation knob. CO₂ enters near the bottom, sinks (1.5× heavier than air), and pushes air up and out through Valve 2
- Screw the valve core back into Valve 2 to seal the chamber
- Detach the inflator from Valve 1. The Presta valve self-closes
- Place in a warm spot (30°C) for 24 hours
- Complete darkness for plants with C3/C4 metabolism
- Under lighting for CAM plants
- Open outdoors or in a well-ventilated area (high CO₂ is dangerous to breathe)
- Repeat after 7 days, then again after another 7 days (3 treatments total, for eggs)
Plant safety notes
I’m not sure of this:
C3/C4 plants (most tropicals, aroids, ferns): treat during the dark period (stomata closed at the night)
CAM plants (cacti, succulents, orchids, hoyas): treat during daylight (stomata closed during the day)
Test on one expendable plant first
.
Shopping list
- tubeless valves https://fr.aliexpress.com/item/1005006614039398.html 3.89€
- CO₂ bike pump https://fr.aliexpress.com/item/1005010155203894.html 5€
- cartridges https://www.amazon.fr/dp/B00DDN4MEQ 10€
- box https://www.amazon.fr/gp/product/B01J5P32ZA 17€ (would be cheaper at IKEA)
Total: 40€ if I can’t find a cheaper box.
Results
First attempt - Cacti with thrips and root mealybugs
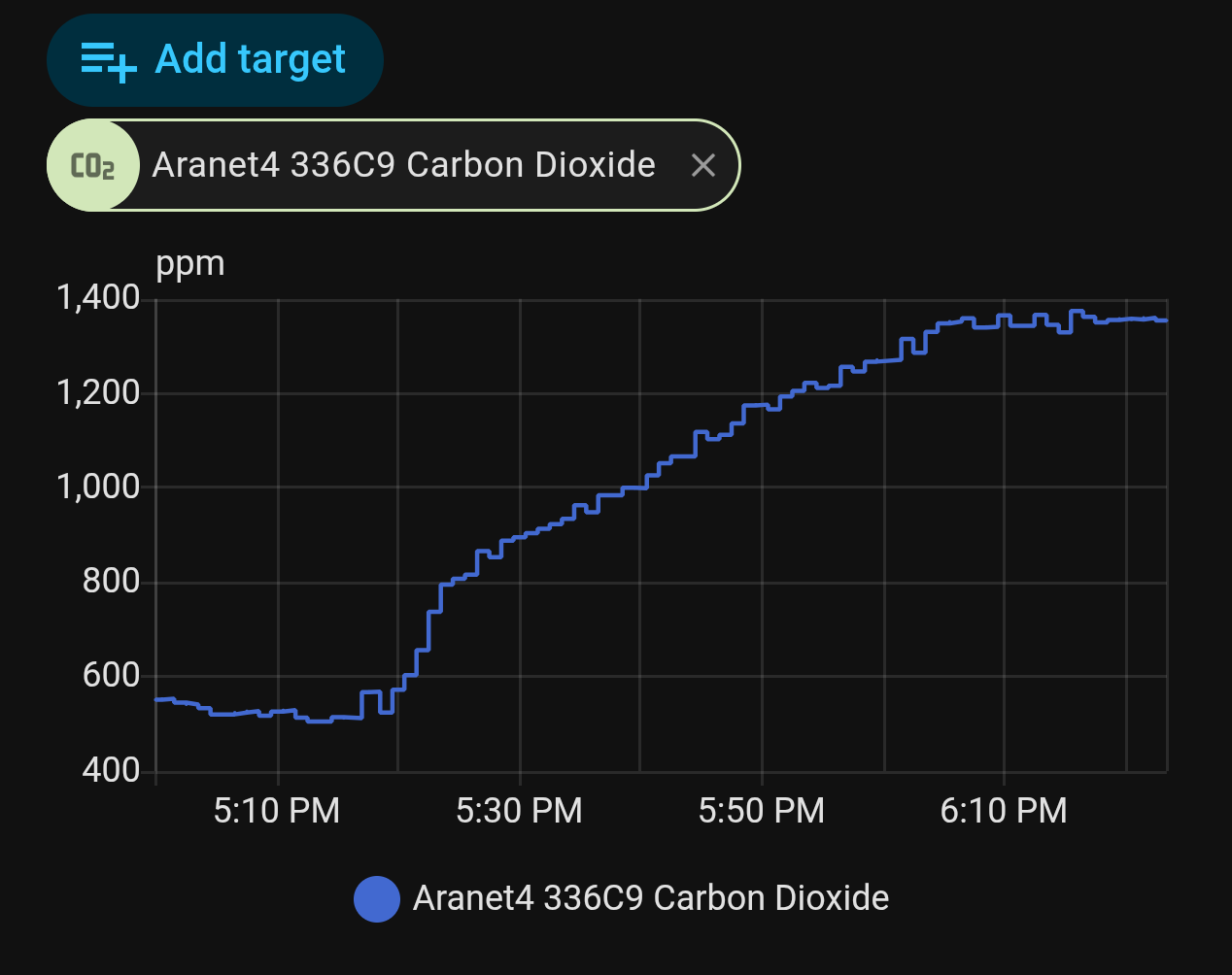
Since all those plants have CAM metabolism, I placed the box in a tent with lights on. My Aranet4 sensor can’t measure above 10000ppm (1% concentration), but I placed it in the tent to see if it leaked.
Concentration rose, and I was concerned that the box was leaking too much to keep the concentration high for 24 hours.
However, when I opened the box, I placed the sensor inside and closed it again (making sure to stay as far as I could to avoid intoxicating myself).
Above the 10000 ppm limit! Since CO2 is denser than air, there might be hope. But without a sensor, there’s no way to be certain…
I spent some time inspecting the plants with my USB microscope, and didn’t see anything moving the next day, a few days after that and two weeks after.
Success? I’m not sure. Getting proof that there are root mealybugs by seeing some crawlers move is easy… but not seeing anything move doesn’t mean there aren’t any live pests still hiding and staying still…
Second attempt
Plants: Tradescantia (some cuttings that started to make roots, and two rooted plants), Coleus cutting and Passiflora cutting.
I’m not expecting the Passiflora cutting to survive and make roots, but it had dozens of thrips so it was a good test to see if they the pests would survive.
This time I added a few more O-rings on the presta valves. It seems to have worked, because the CO2 concentration didn’t rise in the tent.
I added a heat mat under the box, because it thrips mortality (including eggs) in CO2 increases with temperature Seki and Murai (2012b)
Since those plants do not have CAM metabolism, I closed the tent with lights off.
To be continued
(Spoiler: 24 hours later, the bottom of the box was covered with dozens of dead adult thrips!)
Third attempt
April 14, some cactus seedlings and Harrisia cuttings. Lots of root mealybugs in the root balls, and some thrips at least on the Harrisia and maybe on the seedlings.
Results to be updated
Phase 2: Large scale eradication of all pests in my collection
- 2kg CO₂ canister (can be refilled for cheap) or sodastream canisters
(needs adapter for regulators)
- The canister will have to be weighed to know how much CO₂ has been released into the box
- Aquarium CO₂ regulator, maybe something like this https://fr.aliexpress.com/item/1005002734071160.html
- larger box (160L)
- If the box is opaque, might need to install LEDs in the box for CAM plants
- rubber grommets https://fr.aliexpress.com/item/4001045359889.html
- Aquarium tube pipes https://fr.aliexpress.com/item/1005007964433144.html
Will be significantly more expensive because of the large box, the canister and the regulator. Depending on the number of treatments needed, sodastream canister + adapter will have a lower upfront cost, possibly under 100€.
References
Held, D W, R S Gates, D A Potter, and R G Anderson. n.d. “Evaluation of Modified Atmospheres as a Potential Disinfestation Technique for Thrips and Mites in Greenhouses.”
Oyamada, Koichi, and Tamotsu Murai. 2013. “Effect of Fumigation of High Concentration Carbon Dioxide on Two Spotted Spider Mite*,* Tetranychus Urticae Koch (Acari: Tetranychidae) and Strawberry Runner Plant.” Japanese Journal of Applied Entomology and Zoology 57 (4): 249–56. https://doi.org/10.1303/jjaez.2013.249.
↩ 1
Seki, Masao, and Tamotsu Murai. 2012a. “Responses of Five Adult Thrips Species (Thysanoptera; Thripidae) to High-Carbon Dioxide Atmospheres at Different Temperatures.” Applied Entomology and Zoology 47 (2): 125–28. https://doi.org/10.1007/s13355-012-0098-6.
↩ 1
———. 2012b. “Insecticidal Effect of High Carbon Dioxide Atmospheres on Thrips Eggs Oviposited in Plant Tissue.” Applied Entomology and Zoology 47 (4): 433–36. https://doi.org/10.1007/s13355-012-0138-2.
AI disclaimer
- Researched with Claude, Gemini and ChatGPT deep research, but reviewed by me unless noted otherwise
- text mostly written by me
- schematics by gemini